Background
Chiari malformations, types I-IV, refer to a spectrum of congenital hindbrain abnormalities affecting the structural relationships between the cerebellum, brainstem, the upper cervical cord, and the bony cranial base.
History Of The Procedure
Although Cleland described the first cases of Chiari malformation in 1883, the disorder is named after Hans Chiari, an Austrian pathologist, who classified Chiari malformations into types I through III in 1891. Chiari’s colleague, Julius Arnold, made additional contributions to the definition of Chiari II malformation.[1] In his honor, students of Dr. Arnold later named the type II malformation Arnold-Chiari malformation. Other investigators later added the type IV malformation.
Problem
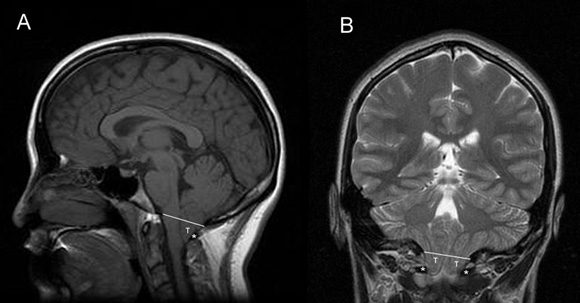
Chiari type I malformation is the most common and the least severe of the spectrum, often diagnosed in adulthood. Its hallmark is caudal displacement of peglike cerebellar tonsils below the level of the foramen magnum, a phenomenon variably referred to as congenital tonsillar herniation, tonsillar ectopia, or tonsillar descent. The resultant impaction of the foramen magnum, compression of the cervicomedullary junction by the ectopic tonsils, and interruption of normal flow of cerebrospinal fluid (CSF) through the region produce the clinical syndrome.
Sagittal and coronal MRI images of Chiari type I malformation. Note descent of cerebellar tonsils (T) below the level of foramen magnum (white line) down to the level of C1 posterior arch (asterisk). Axial MRI image at the level of foramen magnum in Chiari type I malformation. Note crowding of foramen magnum by the ectopic cerebellar tonsils (T) and the medulla (M). Also note the absence of cerebrospinal fluid. Intraoperative photograph of Chiari type 1 malformation showing descent of cerebellar tonsils well below the level of foramen magnum.Chiari type II malformation is less common and more severe, almost invariably associated with myelomeningocele. Because of its greater severity, it becomes symptomatic in infancy or early childhood. Its hallmark is caudal displacement of lower brainstem (medulla, pons, 4th ventricle) through the foramen magnum. Symptoms arise from dysfunction of brainstem and lower cranial nerves.
Chiari type III and IV malformations are exceedingly rare and generally incompatible with life and are, therefore, of scant clinical significance. The type III malformation refers to herniation of cerebellum into a high cervical myelomeningocele, whereas type IV refers to cerebellar agenesis.
Importantly, it is not at all clear that the 4 types of Chiari malformation represent a disease continuum corresponding to a single disorder. The 4 types (particularly types III and IV) are increasingly believed to have different pathogenesis and share little in common other than their names.
This article discusses Chiari type I and II malformations with emphasis on the more common Chiari I malformation.
Frequency
Although Chiari malformation is still listed as a rare disease by the Office of Rare Diseases of the National Institutes of Health, this categorization is based on outdated data from before the MRI era. With routine use of MR imaging, Chiari malformation is discovered with increasing frequency. For Chiari I, prevalence rates of 0.1-0.5% with a slight female predominance are suggested by recent studies.[2] Chiari II is found in all children with myelomeningocele, although less than one-third develop symptoms referable to this malformation.[3]
Based on analysis of familial aggregation, a genetic basis for Chiari I has been suggested.[4] Recent studies suggest linkage to chromosomes 9 and 15.[5] It is hypothesized that Chiari type I originates as a disorder of para-axial mesoderm, which subsequently results in formation of a small posterior fossa. The development of the cerebellum within this small compartment results is overcrowding of the posterior fossa, herniation of the cerebellar tonsils, and impaction of the foramen magnum. This theory is consistent with the observed association of Chiari I and other hereditary mesodermal connective tissue disorders, such as Ehlers-Danlos syndrome.[6]
Theories regarding embryogenesis of Chiari II malformation must take into account its invariable association with myelomeningocele. An attractive theory is the “CSF loss” theory. It is hypothesized that escape of fluid through the open placode in myelomeningocele results in an inadequate stimulus for mesenchymal condensation at the skull base. The disordered and inadequate growth of the posterior fossa results in upward herniation of vermis, downward herniation of brainstem, and distortion of tectum (tectal beaking). Furthermore, collapse of the developing ventricular system because of fluid loss results in associated abnormalities such as agenesis of corpus callosum and enlargement of massa intermedia.
Pathophysiology
Symptoms of Chiari I develop as a result of 3 pathophysiological consequences of the disordered anatomy: (1) compression of medulla and upper spinal cord, (2) compression of cerebellum, and (3) disruption of CSF flow through foramen magnum. Compression of cord and medulla may result in myelopathy and lower cranial nerve and nuclear dysfunction. Compression of cerebellum may result in ataxia, dysmetria, nystagmus, and dysequilibrium. Disruption of CSF flow through foramen magnum probably accounts for the most common symptom, pain.
Accordingly, headache and neck pain in Chiari I are often exacerbated by cough and Valsalva maneuver. Hydrocephalus occurs less frequently. Furthermore, the disordered flow of CSF through foramen magnum may result in formation of syringomyelia and central cord symptoms such as hand weakness and dissociated sensory loss.
The pathophysiology of Chiari II is more complex. Although compressive mechanisms likely play a role, as in Chiari I, additional mechanisms may be operative in Chiari II. Stretching of abnormally oriented cranial nerves is believed to play a role. Chiari II may become acutely symptomatic with shunt malfunction, presumably because hydrocephalus further exacerbates the downward displacement of brainstem and stretching of cranial nerves. It has been suggested that irreversible ischemia of brainstem under tension may be responsible for the poorer prognosis of Chiari II after surgery compared with Chiari I. Furthermore, intrinsic neuroembryological abnormalities in Chiari II are widespread and not limited to the posterior fossa (eg, heterotopias, gyral abnormalities, callosal and thalamic abnormalities, in addition to hydrocephalus and myelomeningocele), further complicating the pathophysiology of this disorder.
Indications
In Chiari I, radiographic presence of tonsillar herniation must correlate with appropriate clinical signs and symptoms before surgical intervention is undertaken. In frankly symptomatic patients, such as those with lower cranial nerve dysfunction, myelopathy, syringomyelia, cerebellar symptoms, or severe post-tussive suboccipital headaches, the decision in favor of surgery is straightforward. Difficulty arises in minimally symptomatic patients or those with equivocal symptoms. CSF flow studies across foramen magnum with phase-contrast cine MRI (see Imaging Studies) may help with surgical decision-making in these cases.
Syringomyelia generally improves or resolves after surgical treatment of Chiari malformation. Rarely is shunting of the Chiari syrinx necessary.
Resolution of syringomyelia (asterisk) after decompression of Chiari I malformation (white arrow).Asymptomatic patients without syringomyelia whose Chiari I malformation has been discovered incidentally on MR imaging do not require surgery. In this group, if the radiographic abnormality appears significant, the patient should be educated about the disorder and asked to seek medical care should symptoms develop in the future.
In Chiari II, when neurological decompensation occurs, the first order of business is to treat hydrocephalus and rule out shunt malfunction. If evidence of brainstem dysfunction is present in spite of well-treated hydrocephalus and a functioning shunt, surgical decompression of Chiari II is undertaken.
Relevant Anatomy
The foramen magnum is an oval-shaped opening in the occipital bone, surrounded anteriorly by the clivus, laterally by the occipital condyles, and posteriorly by the squamous portion of the occipital bone. Normally, only the medulla traverses through the foramen magnum and merges seamlessly with the cervical cord. The lower extension of cisterna magna normally forms a large CSF cushion behind the medulla within the foramen magnum. This CSF cushion is replaced by cerebellar tonsils in Chiari I malformation.
Anatomical knowledge of the foramen magnum dura and its venous sinuses is of particular importance in surgical treatment. The dura that is applied to the inner surface of the squamous portion of occipital bone funnels abruptly into a cylindrical tube at the level of foramen magnum. The squamous occipital dura is bisected vertically by the cerebellar falx. The depth of cerebellar fax between the cerebellar hemispheres diminishes near the foramen magnum. The occipital sinus runs down from the torcula in the trigone formed by the dural leaflets of cerebellar falx and squamous occipital dura. As it approaches the foramen magnum, the occipital sinus divides into two divergent limbs which course laterally around the foramen magnum to join the sigmoid sinuses or the jugular bulbs.
During surgery, dural openings across the foramen magnum are carried out in a Y-shaped fashion in order to avoid the deep part of cerebellar falx and the vertical midline portion of occipital sinus. The two lower limbs of the occipital sinus are transected individually by the two oblique limbs of the Y-shaped incision.
References
- Koehler PJ. Chiari’s description of cerebellar ectopy (1891). With a summary of Cleland’s and Arnold’s contributions and some early observations on neural-tube defects. J Neurosurg. Nov 1991;75(5):823-6. [Medline].
- Speer MC, Enterline DS, Mehltretter L, Hammock P, Joseph J, Dickerson M, et al. Chiari type I malformation with or without syringomyelia: prevalence and genetics. J Genet Couns. 2003;12:297-311.
- Dias M. Myelomeningocoele. In: Choux M, Di Rocco C, Hockley A, Walker M. Pediatric Neurosurgery. London: Churchill Livingston; 1999:33-61.
- Speer MC, George TM, Enterline DS, Franklin A, Wolpert CM, Milhorat TH. A genetic hypothesis for Chiari I malformation with or without syringomyelia. Neurosurg Focus. Mar 15 2000;8(3):E12. [Medline].
- Boyles AL, Enterline DS, Hammock PH, Siegel DG, Slifer SH, Mehltretter L, et al. Phenotypic definition of Chiari type I malformation coupled with high-density SNP genome screen shows significant evidence for linkage to regions on chromosomes 9 and 15. Am J Med Genet A. Dec 15 2006;140(24):2776-85. [Medline].
- Milhorat TH, Bolognese PA, Nishikawa M, McDonnell NB, Francomano CA. Syndrome of occipitoatlantoaxial hypermobility, cranial settling, and chiari malformation type I in patients with hereditary disorders of connective tissue. J Neurosurg Spine. Dec 2007;7(6):601-9. [Medline].
- Greenberg MS. Chiari Malformation. In: Handbook of Neurosurgery. 6. New york: Thieme; 2006:103-109.
- Tubbs RS, Lyerly MJ, Loukas M, Shoja MM, Oakes WJ. The pediatric Chiari I malformation: a review. Childs Nerv Syst. Nov 2007;23(11):1239-50. [Medline].
- Hofkes SK, Iskandar BJ, Turski PA, Gentry LR, McCue JB, Haughton VM. Differentiation between symptomatic Chiari I malformation and asymptomatic tonsilar ectopia by using cerebrospinal fluid flow imaging: initial estimate of imaging accuracy. Radiology. Nov 2007;245(2):532-40. [Medline].
- McGirt MJ, Nimjee SM, Fuchs HE, George TM. Relationship of cine phase-contrast magnetic resonance imaging with outcome after decompression for Chiari I malformations. Neurosurgery. Jul 2006;59(1):140-6; discussion 140-6. [Medline].
- Ulrich Batzdorf. Chiari Malformation. In: M.J. Appuzo. Brain Surgery, Complication Avoidance and Management. 2. New York: Churchill Livingston; 1993.
- James HE. Chiari Malformation Type I. J Neurosurg. Aug 2007;107(2):184. [Medline].
- James HE, Brant A. Treatment of the Chiari malformation with bone decompression without durotomy in children and young adults. Childs Nerv Syst. May 2002;18(5):202-6. [Medline].
- Perrini P, Benedetto N, Tenenbaum R, Di Lorenzo N. Extra-arachnoidal cranio-cervical decompression for syringomyelia associated with Chiari I malformation in adults: technique assessment. Acta Neurochir (Wien). Oct 2007;149(10):1015-22; discussion 1022-3. [Medline].
- Guo F, Wang M, Long J, Wang H, Sun H, Yang B, et al. Surgical management of Chiari malformation: analysis of 128 cases. Pediatr Neurosurg. 2007;43(5):375-81. [Medline].
- Hoffman CE, Souweidane MM. Cerebrospinal fluid-related complications with autologous duraplasty and arachnoid sparing in type I Chiari malformation. Neurosurgery. Mar 2008;62(3 Suppl 1):156-60; discussion 160-1. [Medline].